Biotopin vieraskynäblogi
Impure plasmid DNA may result to erroneous results in downstream processes such as sequencing or even failure in more sensitive processes such as transfection. Thus, different levels of purity are required for specific downstream processes. For example, restriction digestion and cloning can be performed with lower quality plasmid DNA compared to transfection in primary cells.
Purification procedure
Plasmid DNA purification begins with alkaline lysis of bacterial cells. The RNA in the lysis can then be enzymatically digested since the subsequent purification steps seldom distinguish between RNA and DNA. During the third step the cellular proteins and genomic DNA are precipitated and removed via centrifugation. The supernatant contains the plasmid DNA, which is further purified via silica membrane technology or anion exchange chromatography. These methods result in different levels of endpoint plasmid DNA purity and method selection depends on the desired downstream processes.
But what is the difference between them? Products based on silica membrane technology, provide rapid DNA purification from miniscale preps for plasmid DNA of medium grade purity suitable e.g. for sequencing. On the other hand, the products based on anion exchange chromatography to provide scalable method for high yields of ultrapure plasmid DNA especially useful for sensitive processes such as transfection.
Endotoxins
Another point to consider are the endotoxins. More sensitive purposes require plasmid DNA of higher purity or even endotoxin free samples, which must contain less than 0.1 endotoxin unit per µg of DNA (EU/µg of DNA). If simple experiments like sequencing have been planned the silica membrane based methods yield sufficient grade DNA with varying amounts of endotoxins (10-10 000 EU/µg of DNA). For more sensitive downstream processes such as primary cell line transfection, the anion exchange chromatography methods are more suitable. Basic anion exchange chromatography kits are capable of separating DNA and RNA and will only contain 1-10 EU/µg of DNA. They have a yield of 100 µg to 100 mg and with the endotoxin free version will only contain 0.001-0.1 EU/µg of DNA. Thus, transfection even to the most sensitive cells is effective.
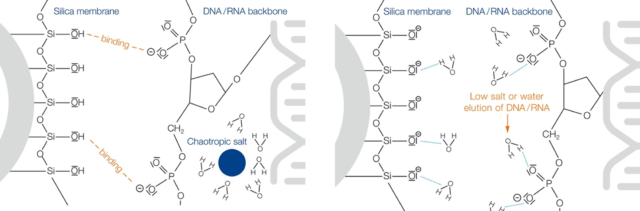
Silica membrane technology
The silica membrane technology was specifically designed for plasmid “minipreps”. The silica membrane technology is based on chaotropic salts which affect the binding of DNA to silica membranes. Chaotropic salts are large, monovalent ions with capabilities to denaturate and disrupt the structure of protein and other macromolecules such as DNA. They display weaker interaction with water molecules compared to water molecules with one another. These features enable the chaotropic salts to perturb the hydrogen bonds in water. The perturbation eliminates the hydration shell around macromolecules like DNA thus increasing their structural freedom. The increasing freedom results in the destabilization of the three-dimensional structure of DNA and enables it to interact with the silica membrane.
As the DNA has bonded to the silica membrane, contaminants may be removed with buffers containing high concentration of chaotropic salts or ethanolic buffers since they eliminate impurities effectively. Then aqueous buffer is used to rehydrate the DNA to break it from the silica membrane thus resulting in elution.
Anion exchange chromatography
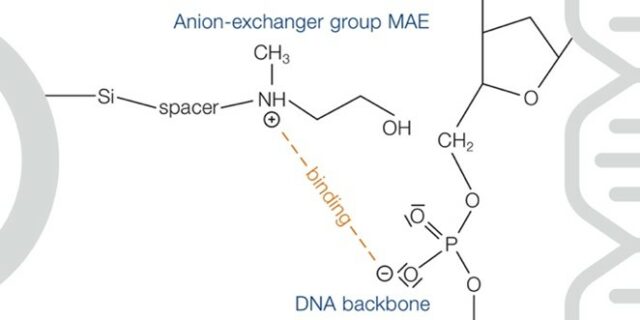
In the anion exchange chromatography the negatively charged DNA in solution interacts with the positively charged molecules in the solid phase anion exchange matrix. Depending on the strength of the negative charge of the DNA, different concentrations of counter ion (often chloride) lead to elution. The solid phase and anion exchange group have been optimized to enable purification of DNA from complex mixtures. Thus, even DNA and RNA may be separated based on their differences in size and charge.
The DNA is eluted under basic conditions to neutralize the positively charge side chain of the anion exchanger, thus resulting in DNA release. After elution, salt removal can be carried out via silica-membrane based syringe filters, which is less laborious and error-prone compared to isopropanol-based DNA precipitation.
The plasmid DNA purified via anion exchange chromatography is suitable for transfection purposes but still contains endotoxins that may decrease transfection efficiency. Since endotoxins are very similar to DNA in charge and size, tailormade solutions are required to separate them via anion exchange chromatography. A patented method which prevents endotoxins from interacting with the anion exchange matrix has been developed come with special buffers to yield plasmid DNA suitable for even the most sensitive downstream processes.
Why choose MN for your plasmid DNA application?
Since the development of kit-based plasmid isolation around 30 years ago, MACHEREY-NAGEL has been a pioneer of supplying fast and highly reliable solutions for the purification of plasmid DNA.
The NucleoSpin® product line using silica membrane technology provides plasmid DNA suitable for most downstream applications. The optimal balance of silica membrane and buffers in NucleoSpin® results in DNA with very long reading-lengths in sequencing reactions. However, manipulation of standard eukaryotic cell lines such as HeLa or HEK cells require a higher purity. For these applications, the NucleoBond® anion exchange chromatography plasmid DNA isolation kits with endotoxin levels between 1–50 EU/μg DNA are recommended. For highly sensitive applications the endotoxin-free kits are highly recommended. These applications include the transfection of precious cell lines, such as primary cells, stem cells or cells growing in low numbers. Macherey-Nagel NucleoBond® endotoxin-free products enable the isolation of plasmid DNA with endotoxin levels below 0.1 EU/μg DNA.
For the best plasmid purification, MACHEREY-NAGEL.
Kirjoittanut: Matti Raitio / Biotop



